-
 Praziquantel Fifty Years on: A Comprehensive Overview of Its Solid State
Praziquantel Fifty Years on: A Comprehensive Overview of Its Solid State -
 Piperacillin/Tazobactam Co-Delivery by Micellar Ionic Conjugate Systems Carrying Pharmaceutical Anions and Encapsulated Drug
Piperacillin/Tazobactam Co-Delivery by Micellar Ionic Conjugate Systems Carrying Pharmaceutical Anions and Encapsulated Drug -
 Lipid-Based Nanotechnology: Liposome
Lipid-Based Nanotechnology: Liposome -
 Chitosan and Cyclodextrins—Versatile Materials Used to Create Drug Delivery Systems for Gastrointestinal Cancers
Chitosan and Cyclodextrins—Versatile Materials Used to Create Drug Delivery Systems for Gastrointestinal Cancers -
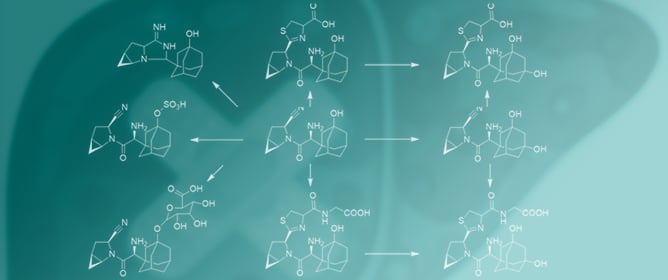 Evaluation of the Drug-Induced Liver Injury Potential of Saxagliptin through Reactive Metabolite Identification in Rats
Evaluation of the Drug-Induced Liver Injury Potential of Saxagliptin through Reactive Metabolite Identification in Rats
Journal Description
Pharmaceutics
Pharmaceutics
is a peer-reviewed, open access journal on the science and technology of pharmaceutics and biopharmaceutics, and is published monthly online by MDPI. The Spanish Society of Pharmaceutics and Pharmaceutical Technology (SEFIG), Pharmaceutical Solid State Research Cluster (PSSRC), Academy of Pharmaceutical Sciences (APS) and Korean Society of Pharmaceutical Sciences and Technology (KSPST) are affiliated with Pharmaceutics and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q1 (Pharmacology & Pharmacy) / CiteScore - Q1 (Pharmaceutical Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.2 days after submission; acceptance to publication is undertaken in 3.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Companion journal: Future Pharmacology
Impact Factor:
5.4 (2022);
5-Year Impact Factor:
6.0 (2022)
Latest Articles
Antibacterial Effect of Ozone on Cariogenic Bacteria and Its Potential Prejudicial Effect on Dentin Bond Strength—An In Vitro Study
Pharmaceutics 2024, 16(5), 614; https://doi.org/10.3390/pharmaceutics16050614 (registering DOI) - 02 May 2024
Abstract
Ozone is increasingly utilized in dental caries treatment due to its antibacterial properties. In a context of limited studies and no consensus on protocols, this research aims to assess ozone’s antibacterial efficacy on cariogenic bacteria and its potential adverse impact on dentin bond
[...] Read more.
Ozone is increasingly utilized in dental caries treatment due to its antibacterial properties. In a context of limited studies and no consensus on protocols, this research aims to assess ozone’s antibacterial efficacy on cariogenic bacteria and its potential adverse impact on dentin bond strength. Streptococcus mutans, Streptococcus sobrinus, Lactobacillus casei, and Actinomyces naeslundii suspensions were exposed to 40 μg/mL of ozone gas and 60 μg/mL of ozonated water (80 s) via a medical ozone generator. Negative and positive control groups (chlorhexidine 2%) were included, and UFC/mL counts were recorded. To examine microtensile bond strength (µTBS), 20 human molars were divided into four groups, and class I cavities were created. After ozone application, samples were restored using an etch-and-rinse and resin composite, then sectioned for testing. The SPSS v. 28 program was used with a significance level of 5%. The µTBS results were evaluated using one-way ANOVA, Tukey HSD, and Games-Howell. Bacterial counts reduced from 106 to 101, but dentin µTBS was significantly impacted by ozone (ANOVA, p < 0.001). Despite ozone’s attractive antibacterial activity, this study emphasizes its detrimental effect on dentin adhesion, cautioning against its use before restorative treatments.
Full article
(This article belongs to the Special Issue Biomaterials and Agents: Pharmaceutical and Biomedical Applications in Dental Research)
►
Show Figures
Open AccessArticle
Ligand- and Structure-Based Virtual Screening Identifies New Inhibitors of the Interaction of the SARS-CoV-2 Spike Protein with the ACE2 Host Receptor
by
Timoteo Delgado-Maldonado, Alonzo González-González, Adriana Moreno-Rodríguez, Virgilio Bocanegra-García, Ana Verónica Martinez-Vazquez, Erick de Jesús de Luna-Santillana, Gerard Pujadas, Guadalupe Rojas-Verde, Edgar E. Lara-Ramírez and Gildardo Rivera
Pharmaceutics 2024, 16(5), 613; https://doi.org/10.3390/pharmaceutics16050613 - 01 May 2024
Abstract
The Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) is a fast-spreading viral pathogen and poses a serious threat to human health. New SARS-CoV-2 variants have been arising worldwide; therefore, is necessary to explore more therapeutic options. The interaction of the viral spike (S)
[...] Read more.
The Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) is a fast-spreading viral pathogen and poses a serious threat to human health. New SARS-CoV-2 variants have been arising worldwide; therefore, is necessary to explore more therapeutic options. The interaction of the viral spike (S) protein with the angiotensin-converting enzyme 2 (ACE2) host receptor is an attractive drug target to prevent the infection via the inhibition of virus cell entry. In this study, Ligand- and Structure-Based Virtual Screening (LBVS and SBVS) was performed to propose potential inhibitors capable of blocking the S receptor-binding domain (RBD) and ACE2 interaction. The best five lead compounds were confirmed as inhibitors through ELISA-based enzyme assays. The docking studies and molecular dynamic (MD) simulations of the selected compounds maintained the molecular interaction and stability (RMSD fluctuations less than 5 Å) with key residues of the S protein. The compounds DRI-1, DRI-2, DRI-3, DRI-4, and DRI-5 efficiently block the interaction between the SARS-CoV-2 spike protein and receptor ACE2 (from 69.90 to 99.65% of inhibition) at 50 µM. The most potent inhibitors were DRI-2 (IC50 = 8.8 µM) and DRI-3 (IC50 = 2.1 µM) and have an acceptable profile of cytotoxicity (CC50 > 90 µM). Therefore, these compounds could be good candidates for further SARS-CoV-2 preclinical experiments.
Full article
(This article belongs to the Special Issue Uncovering the Newest Research in Antiviral-Drugs Development and Potential of Combined Therapy)
►▼
Show Figures
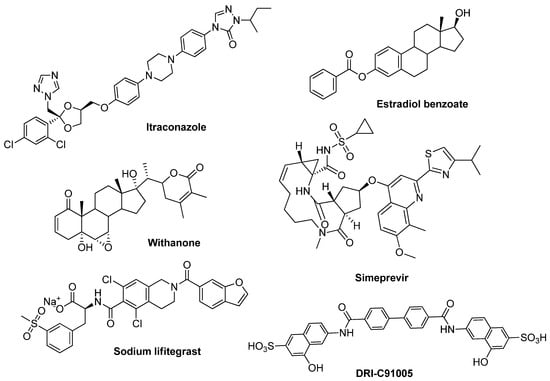
Figure 1
Open AccessArticle
Natural Phytochemical and Visible Light at Different Wavelengths Show Synergistic Antibacterial Activity against Staphylococcus aureus
by
Jae-Young Jeong and You-Jin Hwang
Pharmaceutics 2024, 16(5), 612; https://doi.org/10.3390/pharmaceutics16050612 - 01 May 2024
Abstract
As the risk of antibiotic-resistant bacteria increases, interest in non-antibiotic treatment is also increasing. Among the methods used in non-antibiotic therapy, natural antibiotics such as essential oils have disadvantages such as low efficiency. In the case of phototherapy, the light used for antibacterial
[...] Read more.
As the risk of antibiotic-resistant bacteria increases, interest in non-antibiotic treatment is also increasing. Among the methods used in non-antibiotic therapy, natural antibiotics such as essential oils have disadvantages such as low efficiency. In the case of phototherapy, the light used for antibacterial activities has low penetration into the human body because of its short wavelength, making it of low medical utility. To solve this problem, this study aimed to determine conditions for enhancing the antibacterial activity of natural phytochemicals and visible light. Four natural phytochemical extracts that showed high antibacterial properties in previous studies were analyzed. Synergistic effects on antibacterial activity and cytotoxicity were determined when natural phytochemical extracts and visible light were simultaneously used. As a result, it was confirmed that the antibacterial activity increased by four times when Sanguisorba officinalis L. was irradiated with 465 nm for 10 min and 520 nm for 40 min, and Uncaria gambir Roxb. was irradiated with 465 nm for 10 min and 520 nm for 60 min compared to when Sanguisorba officinalis L. and Uncaria gambir Roxb. were used alone. The synergistic effect on antibacterial activity was independent of the absorption peak of the natural phytochemical extracts. In addition, in the case of natural phytochemical extracts with improved antibacterial activity, it was confirmed that the improvement of antibacterial activity was increased in inverse proportion to the light irradiation wavelength and in proportion to the light irradiation time. The antibacterial activity was enhanced regardless of antibiotic resistance. In the case of cytotoxicity, it was confirmed that there was no toxicity to A549 cells when treated with 465 nm, the shortest wavelength among the natural phytochemical extracts. These results show how to replace blue light, which has been underutilized due to its low transmittance and cytotoxicity. They also demonstrate the high medical potential of using natural phytochemical and visible light as a combination therapy.
Full article
(This article belongs to the Special Issue Natural Products in Photodynamic Therapy)
►▼
Show Figures
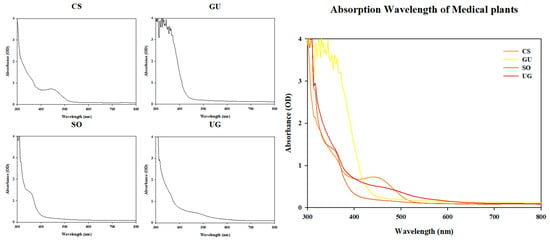
Figure 1
Open AccessArticle
The Role of a Cholecystokinin Receptor Antagonist in the Management of Chronic Pancreatitis: A Phase 1 Trial
by
Victor Ciofoaia, Wenqiang Chen, Bakain W. Tarek, Martha Gay, Narayan Shivapurkar and Jill P. Smith
Pharmaceutics 2024, 16(5), 611; https://doi.org/10.3390/pharmaceutics16050611 - 30 Apr 2024
Abstract
Chronic pancreatitis (CP) is a rare but debilitating condition with an 8-fold increased risk of developing pancreatic cancer. In addition to the symptoms that come from the loss of endocrine and exocrine function in CP, the management of chronic pain is problematic. We
[...] Read more.
Chronic pancreatitis (CP) is a rare but debilitating condition with an 8-fold increased risk of developing pancreatic cancer. In addition to the symptoms that come from the loss of endocrine and exocrine function in CP, the management of chronic pain is problematic. We previously showed that the CCK-receptor antagonist called proglumide could decrease inflammation, acinar-ductal metaplasia, and fibrosis in murine models of CP. We hypothesized that proglumide would be safe and diminish pain caused by CP. A Phase 1 open-labeled safety study was performed in subjects with clinical and radiographic evidence of CP with moderate to severe pain. After a 4-week observation period, the subjects were treated with proglumide in 400 mg capsules three times daily (1200 mg per day) by mouth for 12 weeks, followed by 4 weeks of observation after discontinuation for safety. The results of three pain surveys (Numeric Rating Scale, COMPAT-SF, and NIH PROMIS) showed that the patients had significantly less pain after 12 weeks of proglumide compared to the pre-treatment observation phase. Of the eight subjects in this study, two experienced nausea and diarrhea with proglumide. These side effects resolved in one subject with doses reduced to 800 mg per day. No abnormalities were noted in the blood chemistries. A blood microRNA blood biomarker panel that corresponded to pancreatic inflammation and fibrosis showed significant improvement. We conclude that proglumide is safe and well tolerated in most subjects with CP at a dose of 1200 mg per day. Furthermore, proglumide therapy may have a beneficial effect by decreasing pain associated with CP.
Full article
(This article belongs to the Special Issue New Pharmaceutical Targets to Counteract Chronic Inflammation)
Open AccessArticle
General Direct Anticancer Effects of Deer Growing Antler Extract in Several Tumour Cell Lines, and Immune System-Mediated Effects in Xenograft Glioblastoma
by
Alessandra Rossetti, Louis Chonco, Nicolas Alegría, Veronica Zelli, Andrés J. García, Carmen Ramírez-Castillejo, Alessandra Tessitore, Carlos de Cabo, Tomás Landete-Castillejos and Claudio Festuccia
Pharmaceutics 2024, 16(5), 610; https://doi.org/10.3390/pharmaceutics16050610 - 30 Apr 2024
Abstract
Deer antlers are the fastest growing tissue. Because they are based on proto-oncogenes, to avoid the risk of cancer, antlers evolved strong anticancer mechanisms, and thus their extract (DVA) is effective also against the few human tumours studied so far. We assessed whether
[...] Read more.
Deer antlers are the fastest growing tissue. Because they are based on proto-oncogenes, to avoid the risk of cancer, antlers evolved strong anticancer mechanisms, and thus their extract (DVA) is effective also against the few human tumours studied so far. We assessed whether DVA is a general anticancer compound by testing the direct effects in cells of different tumours: glioblastoma (GBM; lines U87MG and U251), colorectal (CRC; lines DLD-1, HT-29, SW480, and SW620), breast cancer (BRCA; lines MCF7, SKBR3, and PA00), and leukaemia (THP-1). DVA reduced the viability of tumours but not healthy cells (NHC; lines 293T and HaCaT). Mobility decreased at least for the longest test (72 h). Intraperitoneal/oral 200 mg DVA/kg administration in GBM xenograft mice for 28 d reduced tumour weight by 66.3% and 61.4% respectively, and it also reduced spleen weight (43.8%). In addition, tumours treated with DVA showed symptoms of liquefactive necrosis. Serum cytokines showed DVA up-regulated factors related to tumour fighting and down-regulated those related to inducing immune tolerance to the tumour. DVA shows general anticancer effects in the lines tested and, in GBM mice, also strong indirect effects apparently mediated by the immune system. DVA may contain a future anticancer medicine without secondary effects.
Full article
(This article belongs to the Topic Antitumor Activity of Natural Products and Related Compounds, 2nd Volume)
Open AccessReview
Nonintuitive Immunogenicity and Plasticity of Alpha-Synuclein Conformers: A Paradigm for Smart Delivery of Neuro-Immunotherapeutics
by
Amos Abioye, Damilare Akintade, James Mitchell, Simisade Olorode and Adeboye Adejare
Pharmaceutics 2024, 16(5), 609; https://doi.org/10.3390/pharmaceutics16050609 - 30 Apr 2024
Abstract
: Despite the extensive research successes and continuous developments in modern medicine in terms of diagnosis, prevention, and treatment, the lack of clinically useful disease-modifying drugs or immunotherapeutic agents that can successfully treat or prevent neurodegenerative diseases is an ongoing challenge. To date,
[...] Read more.
: Despite the extensive research successes and continuous developments in modern medicine in terms of diagnosis, prevention, and treatment, the lack of clinically useful disease-modifying drugs or immunotherapeutic agents that can successfully treat or prevent neurodegenerative diseases is an ongoing challenge. To date, only one of the 244 drugs in clinical trials for the treatment of neurodegenerative diseases has been approved in the past decade, indicating a failure rate of 99.6%. In corollary, the approved monoclonal antibody did not demonstrate significant cognitive benefits. Thus, the prevalence of neurodegenerative diseases is increasing rapidly. Therefore, there is an urgent need for creative approaches to identifying and testing biomarkers for better diagnosis, prevention, and disease-modifying strategies for the treatment of neurodegenerative diseases. Overexpression of the endogenous α-synuclein has been identified as the driving force for the formation of the pathogenic α-synuclein (α-Syn) conformers, resulting in neuroinflammation, hypersensitivity, endogenous homeostatic responses, oxidative dysfunction, and degeneration of dopaminergic neurons in Parkinson’s disease (PD). However, the conformational plasticity of α-Syn proffers that a certain level of α-Syn is essential for the survival of neurons. Thus, it exerts both neuroprotective and neurotoxic (regulatory) functions on neighboring neuronal cells. Furthermore, the aberrant metastable α-Syn conformers may be subtle and difficult to detect but may trigger cellular and molecular events including immune responses. It is well documented in literature that the misfolded α-Syn and its conformers that are released into the extracellular space from damaged or dead neurons trigger the innate and adaptive immune responses in PD. Thus, in this review, we discuss the nonintuitive plasticity and immunogenicity of the α-Syn conformers in the brain immune cells and their physiological and pathological consequences on the neuroimmune responses including neuroinflammation, homeostatic remodeling, and cell-specific interactions that promote neuroprotection in PD. We also critically reviewed the novel strategies for immunotherapeutic delivery interventions in PD pathogenesis including immunotherapeutic targets and potential nanoparticle-based smart drug delivery systems. It is envisioned that a greater understanding of the nonintuitive immunogenicity of aberrant α-Syn conformers in the brain’s microenvironment would provide a platform for identifying valid therapeutic targets and developing smart brain delivery systems for clinically effective disease-modifying immunotherapeutics that can aid in the prevention and treatment of PD in the future.
Full article
(This article belongs to the Special Issue Application of Nanomedicine in Immunotherapy: Recent Advances and Prospects, 2nd Edition)
Open AccessArticle
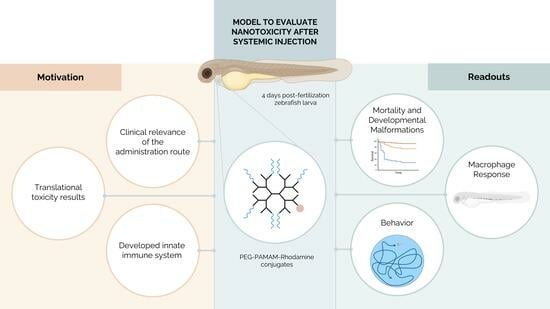
Biological Response Following the Systemic Injection of PEG–PAMAM–Rhodamine Conjugates in Zebrafish
by
Beatriz Custódio, Patrícia Carneiro, Joana Marques, Victoria Leiro, Ana M. Valentim, Mafalda Sousa, Sofia D. Santos, José Bessa and Ana P. Pêgo
Pharmaceutics 2024, 16(5), 608; https://doi.org/10.3390/pharmaceutics16050608 - 30 Apr 2024
Abstract
Numerous therapeutic and diagnostic approaches used within a clinical setting depend on the administration of compounds via systemic delivery. Biomaterials at the nanometer scale, as dendrimers, act as delivery systems by improving cargo bioavailability, circulation time, and the targeting of specific tissues. Although
[...] Read more.
Numerous therapeutic and diagnostic approaches used within a clinical setting depend on the administration of compounds via systemic delivery. Biomaterials at the nanometer scale, as dendrimers, act as delivery systems by improving cargo bioavailability, circulation time, and the targeting of specific tissues. Although evaluating the efficacy of pharmacological agents based on nanobiomaterials is crucial, conducting toxicological assessments of biomaterials is essential for advancing clinical translation. Here, a zebrafish larvae model was explored to assess the biocompatibility of poly(amido amine) (PAMAM), one of the most exploited dendrimers for drug delivery. We report the impact of a systemic injection of polyethylene glycol (PEG)-modified G4 PAMAM conjugated with rhodamine (Rho) as a mimetic drug (PEG–PAMAM–Rho) on survival, animal development, inflammation, and neurotoxicity. A concentration- and time-dependent effect was observed on mortality, developmental morphology, and innate immune system activation (macrophages). Significant effects in toxicological indicators were reported in the highest tested concentration (50 mg/mL PEG–PAMAM–Rho) as early as 48 h post-injection. Additionally, a lower concentration of PEG–PAMAM–Rho (5 mg/mL) was found to be safe and subsequently tested for neurotoxicity through behavioral assays. In accordance, no significative signs of toxicity were detected. In conclusion, the dose response of the animal was assessed, and the safe dosage for future use in theragnostics was defined. Additionally, new methodologies were established that can be adapted to further studies in toxicology using other nanosystems for systemic delivery.
Full article
(This article belongs to the Special Issue Nano-Based Drug Delivery System: Recent Developments and Future Prospects)
►▼
Show Figures
Graphical abstract
Open AccessArticle
In Vitro Human Liver Model for Toxicity Assessment with Clinical and Preclinical Instrumentation
by
Eneko Madorran, Lidija Kocbek Šaherl, Mateja Rakuša and Miha Munda
Pharmaceutics 2024, 16(5), 607; https://doi.org/10.3390/pharmaceutics16050607 - 29 Apr 2024
Abstract
The existing in vitro toxicological models lack translational potential, which makes difficult the application of gathered information to clinical usage. To tackle this issue, we built a model with four different types of primary liver cells: hepatic sinusoidal endothelial cells, hepatic stellate cells,
[...] Read more.
The existing in vitro toxicological models lack translational potential, which makes difficult the application of gathered information to clinical usage. To tackle this issue, we built a model with four different types of primary liver cells: hepatic sinusoidal endothelial cells, hepatic stellate cells, Kupffer cells and hepatocytes. We cultured them in different combinations of composition and volumes of cell medium, hepatocyte proportions of total cells and additions of extracellular matrixes. We added rifampicin (RIF), ibuprofen (IBU) and 5-fluorouracil (5-FU) to this model and observed the microanatomy and physiology changes for a week with preclinical and clinical instruments. Among the different model configurations, we selected the feature combination of the in vitro model that had similar biomarker values to those measured in clinical diagnostics. When we exposed the selected model configuration to RIF, IBU and 5-FU, we observed similar glucose, triglyceride and albumin dynamics as in vivo (from clinical data). Therefore, we have built an in vitro liver model that resembles the liver microenvironment, and we have analysed it with clinical instrumentation to facilitate data translation. Furthermore, during these observations, we found that Kupffer and LSEC cells are suitable candidates for the search for clinical diagnostic markers of liver function.
Full article
(This article belongs to the Special Issue Alternative Model-Based Translational Drug Development)
Open AccessArticle
Valorisation of Raspberry Seeds in Cosmetic Industry-Green Solutions
by
Ivanka Ćirić, Dragana Dabić Zagorac, Milica Sredojević, Milica Fotirić Akšić, Biljana Rabrenović, Stevan Blagojević and Maja Natić
Pharmaceutics 2024, 16(5), 606; https://doi.org/10.3390/pharmaceutics16050606 - 29 Apr 2024
Abstract
The fruit processing industry generates large quantities of by-products well known to be rich in bioactive compounds with numerous nutritional properties and beneficial effects for human health. We developed a strategy to valorise raspberry seeds and obtain valuable ingredients with potential application in
[...] Read more.
The fruit processing industry generates large quantities of by-products well known to be rich in bioactive compounds with numerous nutritional properties and beneficial effects for human health. We developed a strategy to valorise raspberry seeds and obtain valuable ingredients with potential application in cosmetic skincare formulas. Cold press extraction technology was applied to extract oil, and the remaining defatted raspberry seed cake was treated with three proline based deep eutectic solvents (DES) to extract polyphenols. The most potent was proline/citric acid extract, with free and total ellagic acid content (52.4 mg/L and 86.4 mg/L), total phenolic content (TPC, 550.1 mg GAE/L) and radical scavenging activity (RSA, 4742.7 mmol TE/L). After the direct mixing of the extract and after encapsulation with starch as a carrier, the skincare emulsion and microemulsion were characterised by irritation potential (Zein test), transepidermal water loss (TEWL), red blood cell (RBC), and DPPH antioxidant test. The resulting preparations were of improved quality in comparison to the control hand cream, with a low skin irritation effect, lower TEWL, and higher antioxidant potential. This work complies with circular economy principles and green technology standards, and represents the efficient model on how to reuse natural resources through waste minimization.
Full article
(This article belongs to the Special Issue Skin Care Products for Healthy and Diseased Skin)
Open AccessArticle
Again and Again—Survival of Candida albicans in Urine Containing Antifungals
by
Nevio Facchini, Lukas Wernli, Malte Rieken, Gernot Bonkat, Dieter Wirz and Olivier Braissant
Pharmaceutics 2024, 16(5), 605; https://doi.org/10.3390/pharmaceutics16050605 - 29 Apr 2024
Abstract
Background: Relapse of Candida albicans urinary tract infection (UTI) is frequent despite appropriate treatment, as commonly used antifungals such fluconazole and flucytosine are only fungistatics. To improve treatment of Candida UTI and decrease relapses, understanding the long-term metabolic activity and survival of C.
[...] Read more.
Background: Relapse of Candida albicans urinary tract infection (UTI) is frequent despite appropriate treatment, as commonly used antifungals such fluconazole and flucytosine are only fungistatics. To improve treatment of Candida UTI and decrease relapses, understanding the long-term metabolic activity and survival of C. albicans in urine containing antifungals at minimal inhibitory concentration (MIC) is needed. Methods: we monitored the survival, metabolic activity and consumption of glucose and proteins by C. albicans using conventional methods and isothermal microcalorimetry (IMC). We also investigated the influence of dead Candida cells on the growth of their living counterparts. Results: For 33 days, weak activity was observed in samples containing antifungals in which C. albicans growth rate was reduced by 48%, 60% and 88%, and the lag increased to 172 h, 168 h and 6 h for amphotericin, flucytosine and fluconazole, respectively. The metabolic activity peaks corresponded to the plate counts but were delayed compared to the exhaustion of resources. The presence of dead cells promoted growth in artificial urine, increasing growth rate and reducing lag in similar proportions. Conclusions: Even with antifungal treatment, C. albicans relapses are possible. The low metabolic activity of surviving cells leading to regrowth and chlamydospore formation possibly supported by autophagy are likely important factors in relapses.
Full article
(This article belongs to the Special Issue Recent Advances in Prevention and Treatment of Infectious Diseases)
Open AccessArticle
Characterization of Surfactant Spheroidal Micelle Structure for Pharmaceutical Applications: A Novel Analytical Framework
by
Liberato De Caro, Thibaud Stoll, Arnaud Grandeury, Fabia Gozzo and Cinzia Giannini
Pharmaceutics 2024, 16(5), 604; https://doi.org/10.3390/pharmaceutics16050604 - 29 Apr 2024
Abstract
We introduce an innovative theoretical framework tailored for the analysis of Pair Distribution Function (PDF) data derived from Small-Angle X-ray Scattering (SAXS) measurements of core-shell micelles. The new approach involves the exploitation of the first derivative of the PDF and the derivation of
[...] Read more.
We introduce an innovative theoretical framework tailored for the analysis of Pair Distribution Function (PDF) data derived from Small-Angle X-ray Scattering (SAXS) measurements of core-shell micelles. The new approach involves the exploitation of the first derivative of the PDF and the derivation of analytical equations to solve the core-shell micelle structure under the hypothesis of a spheroidal shape. These analytical equations enable us to determine the micelle’s aggregation number, degree of ellipticity, and contrast in electron density between the core-shell and shell-buffer regions after having determined the whole micelle size and its shell size from the analysis of the first derivative of the PDF. We have formulated an overdetermined system of analytical equations based on the unknowns that characterize the micelle structure. This allows us to establish a Figure of Merit, which is utilized to identify the most reliable solution within the system of equations.
Full article
Open AccessReview
Application of Nanomaterial-Based Sonodynamic Therapy in Tumor Therapy
by
Nan Yang, Jianmin Li, Shujie Yu, Guoyu Xia, Dingyang Li, Longlong Yuan, Qingluo Wang, Lijun Ding, Zhongxiong Fan and Jinyao Li
Pharmaceutics 2024, 16(5), 603; https://doi.org/10.3390/pharmaceutics16050603 - 29 Apr 2024
Abstract
Sonodynamic therapy (SDT) has attracted significant attention in recent years as it is an innovative approach to tumor treatment. It involves the utilization of sound waves or ultrasound (US) to activate acoustic sensitizers, enabling targeted drug release for precise tumor treatment. This review
[...] Read more.
Sonodynamic therapy (SDT) has attracted significant attention in recent years as it is an innovative approach to tumor treatment. It involves the utilization of sound waves or ultrasound (US) to activate acoustic sensitizers, enabling targeted drug release for precise tumor treatment. This review aims to provide a comprehensive overview of SDT, encompassing its underlying principles and therapeutic mechanisms, the applications of nanomaterials, and potential synergies with combination therapies. The review begins by introducing the fundamental principle of SDT and delving into the intricate mechanisms through which it facilitates tumor treatment. A detailed analysis is presented, outlining how SDT effectively destroys tumor cells by modulating drug release mechanisms. Subsequently, this review explores the diverse range of nanomaterials utilized in SDT applications and highlights their specific contributions to enhancing treatment outcomes. Furthermore, the potential to combine SDT with other therapeutic modalities such as photothermal therapy (PTT) and chemotherapy is discussed. These combined approaches aim to synergistically improve therapeutic efficacy while mitigating side effects. In conclusion, SDT emerges as a promising frontier in tumor treatment that offers personalized and effective treatment options with the potential to revolutionize patient care. As research progresses, SDT is poised to play a pivotal role in shaping the future landscape of oncology by providing patients with a broader spectrum of efficacious and tailored treatment options.
Full article
(This article belongs to the Special Issue Ultrasound Assisted in Tumor Immunotherapy)
Open AccessArticle
A Sensitive Assay for Unbound Docetaxel Using Ultrafiltration plus HPLC-MS and Its Application to a Clinical Study
by
David Wang, Natalie Hughes-Medlicott, Lilian Klingler, Yi Wang, Noelyn Hung, Stephen Duffull, Tak Hung, Paul Glue, Albert Qin, Rudolf Kwan, Wing-Kai Chan and Christopher Jackson
Pharmaceutics 2024, 16(5), 602; https://doi.org/10.3390/pharmaceutics16050602 - 29 Apr 2024
Abstract
Introduction: Docetaxel, a taxane used in the treatment of solid tumours, exerts pharmacological activity when in its unbound form. We report a sensitive assay to quantify unbound docetaxel after oral administration of docetaxel plus encequidar (oDox+E). Unbound drug quantification is important due to
[...] Read more.
Introduction: Docetaxel, a taxane used in the treatment of solid tumours, exerts pharmacological activity when in its unbound form. We report a sensitive assay to quantify unbound docetaxel after oral administration of docetaxel plus encequidar (oDox+E). Unbound drug quantification is important due to its direct correlation with drug-related toxicity and therapeutic efficacy. We improve on the sensitivity of current assay methods and demonstrate the utility of the assay on a novel formulation of oral docetaxel. Methods: Ultrafiltration followed by high-performance liquid chromatography and tandem mass spectrometry (HPLC-MS/MS) was utilized. Long-term stability, precision, accuracy, and recovery experiments were conducted to validate the assay. Additionally, patient samples from a Phase I dose-escalation pharmacokinetic study were analyzed using the developed assay. Results: The assay method exhibited long-term stability with an observed change between 0.8 and 6.9% after 131 days of storage at −60 °C. Precision and accuracy quality controls met the FDA acceptance criteria. An average recovery of 88% was obtained. Patient sample analysis demonstrated successful implementation of the assay. Conclusion: A validated sensitive assay was developed with an LLOQ of 0.084 ng/mL using 485 µL of human plasma. The sensitivity of the assay allowed quantification of unbound docetaxel concentrations in an early-phase oDox+E clinical study to compare it against IV docetaxel using pharmacokinetic modelling. Successful development of oDox+E represents an opportunity to replace the current IV docetaxel regimen with an oral regimen with lower cost, decreased side effects, and improve patient quality of life and experience.
Full article
(This article belongs to the Special Issue Development of Novel Tumor-Targeting Nanoparticles, 2nd Edition)
►▼
Show Figures
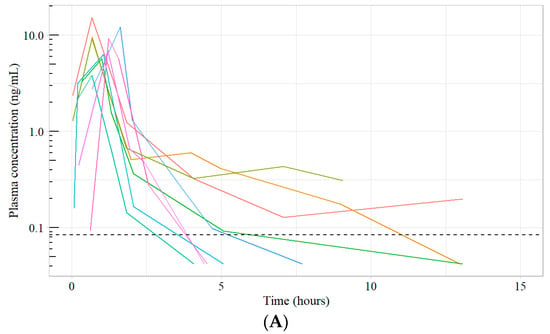
Figure 1
Open AccessArticle
Enhanced In Vitro Antiviral Activity of Ivermectin-Loaded Nanostructured Lipid Carriers against Porcine Epidemic Diarrhea Virus via Improved Intracellular Delivery
by
Xiaolin Xu, Shasha Gao, Qindan Zuo, Jiahao Gong, Xinhao Song, Yongshi Liu, Jing Xiao, Xiaofeng Zhai, Haifeng Sun, Mingzhi Zhang, Xiuge Gao and Dawei Guo
Pharmaceutics 2024, 16(5), 601; https://doi.org/10.3390/pharmaceutics16050601 - 29 Apr 2024
Abstract
Porcine epidemic diarrhea virus (PEDV) is an acute enteric coronavirus, inducing watery diarrhea and high mortality in piglets, leading to huge economic losses in global pig industry. Ivermectin (IVM), an FDA-approved antiparasitic agent, is characterized by high efficacy and wide applicability. However, the
[...] Read more.
Porcine epidemic diarrhea virus (PEDV) is an acute enteric coronavirus, inducing watery diarrhea and high mortality in piglets, leading to huge economic losses in global pig industry. Ivermectin (IVM), an FDA-approved antiparasitic agent, is characterized by high efficacy and wide applicability. However, the poor bioavailability limits its application. Since the virus is parasitized inside the host cells, increasing the intracellular drug uptake can improve antiviral efficacy. Hence, we aimed to develop nanostructured lipid carriers (NLCs) to enhance the antiviral efficacy of IVM. The findings first revealed the capacity of IVM to inhibit the infectivity of PEDV by reducing viral replication with a certain direct inactivation effect. The as-prepared IVM-NLCs possessed hydrodynamic diameter of 153.5 nm with a zeta potential of −31.5 mV and high encapsulation efficiency (95.72%) and drug loading (11.17%). IVM interacted with lipids and was enveloped in lipid carriers with an amorphous state. Furthermore, its encapsulation in NLCs could enhance drug internalization. Meanwhile, IVM-NLCs inhibited PEDV proliferation by up to three orders of magnitude in terms of viral RNA copies, impeding the accumulation of reactive oxygen species and mitigating the mitochondrial dysfunction caused by PEDV infection. Moreover, IVM-NLCs markedly decreased the apoptosis rate of PEDV-induced Vero cells. Hence, IVM-NLCs showed superior inhibitory effect against PEDV compared to free IVM. Together, these results implied that NLCs is an efficient delivery system for IVM to improve its antiviral efficacy against PEDV via enhanced intracellular uptake.
Full article
(This article belongs to the Special Issue Lipid/Polymer-Based Drug Delivery Systems)
►▼
Show Figures
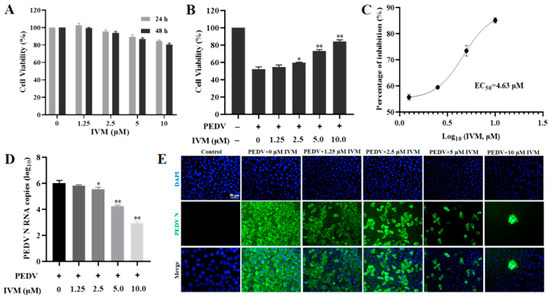
Figure 1
Open AccessArticle
Linseed Oil-Based Oleogel Vehicles for Hydrophobic Drug Delivery—Physicochemical and Applicative Properties
by
Sonia Kudłacik-Kramarczyk, Anna Drabczyk, Alicja Przybyłowicz and Marcel Krzan
Pharmaceutics 2024, 16(5), 600; https://doi.org/10.3390/pharmaceutics16050600 - 29 Apr 2024
Abstract
In this study, a methodology for synthesizing oleogels based on linseed oil and emulsifiers, such as beeswax and Tween 20 and Tween 80, was developed. Linseed oil served as the main oil phase, while beeswax acted as a gelling and emulsifying agent. Tween
[...] Read more.
In this study, a methodology for synthesizing oleogels based on linseed oil and emulsifiers, such as beeswax and Tween 20 and Tween 80, was developed. Linseed oil served as the main oil phase, while beeswax acted as a gelling and emulsifying agent. Tween compounds are non-ionic surfactants composed of hydrophobic and hydrophilic parts, allowing for the formation of a stable system with promising properties. Surface wetting analysis of the obtained oleogels, FT-IR spectroscopy, and determination of relative and absolute humidity over time, as well as optical microscope analysis and rheological analysis of the obtained oleogels, were conducted as part of the research. The results indicate that increasing the amount of Tween 20 decreases the hydrophilicity of the oleogel, while Tween 80 exhibits the opposite effect. Surface energy analysis suggests that a higher content of Tween 20 may lead to a reduction in the surface energy of the oleogels, which may indicate greater material stability. Changes in relative humidity and FT-IR spectral analysis confirm the influence of emulsifiers on the presence of characteristic functional groups in the structure of the oleogels. Additionally, microscopic analysis suggests that an emulsifier with a longer hydrophobic tail leads to a denser material structure.
Full article
(This article belongs to the Special Issue Functional Biomaterials in Biomedical Applications)
►▼
Show Figures
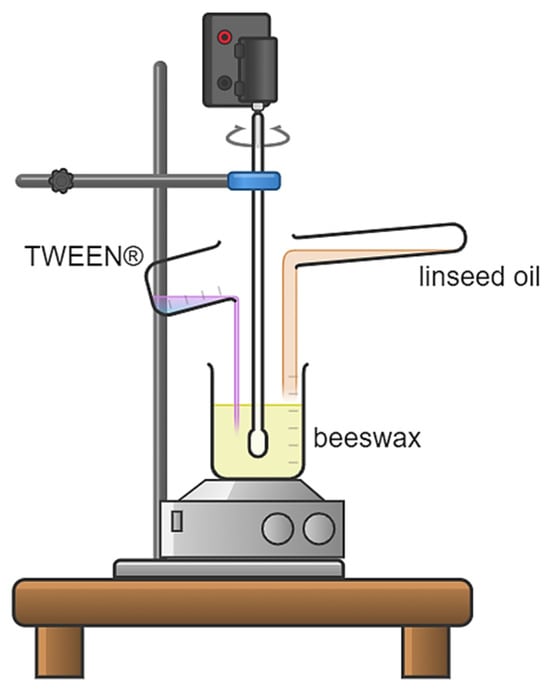
Figure 1
Open AccessArticle
Development of 5-Fluorouracil/pH-Responsive Adjuvant-Embedded Extracellular Vesicles for Targeting αvβ3 Integrin Receptors in Tumors
by
Jiseung Kim, Eunsol Lee and Eun Seong Lee
Pharmaceutics 2024, 16(5), 599; https://doi.org/10.3390/pharmaceutics16050599 - 29 Apr 2024
Abstract
To selectively target and treat murine melanoma B16BL6 tumors expressing αvβ3 integrin receptors, we engineered tumor-specific functional extracellular vesicles (EVs) tailored for the targeted delivery of antitumor drugs. This objective was achieved through the incorporation of a pH-responsive adjuvant, cyclic
[...] Read more.
To selectively target and treat murine melanoma B16BL6 tumors expressing αvβ3 integrin receptors, we engineered tumor-specific functional extracellular vesicles (EVs) tailored for the targeted delivery of antitumor drugs. This objective was achieved through the incorporation of a pH-responsive adjuvant, cyclic arginine-glycine-aspartic acid peptide (cRGD, serving as a tumor-targeting ligand), and 5-fluorouracil (5-FU, employed as a model antitumor drug). The pH-responsive adjuvant, essential for modulating drug release, was synthesized by chemically conjugating 3-(diethylamino)propylamine (DEAP) to deoxycholic acid (DOCA, a lipophilic substance capable of integrating into EVs’ membranes), denoted as DEAP-DOCA. The DOCA, preactivated using N-(2-aminoethyl)maleimide (AEM), was chemically coupled with the thiol group of the cRGD-DOCA through the thiol–maleimide click reaction, resulting in the formation of cRGD-DOCA. Subsequently, DEAP-DOCA, cRGD-DOCA, and 5-FU were efficiently incorporated into EVs using a sonication method. The resulting tumor-targeting EVs, expressing cRGD ligands, demonstrated enhanced in vitro/in vivo cellular uptake specifically for B16BL6 tumors expressing αvβ3 integrin receptors. The ionization characteristics of the DEAP in DEAP-DOCA induced destabilization of the EVs membrane at pH 6.5 through protonation of the DEAP substance, thereby expediting 5-FU release. Consequently, an improvement in the in vivo antitumor efficacy was observed for B16BL6 tumors. Based on these comprehensive in vitro/in vivo findings, we anticipate that this EV system holds substantial promise as an exceptionally effective platform for antitumor therapeutic delivery.
Full article
(This article belongs to the Special Issue Extracellular Vesicle-Based Drug Delivery Systems)
►▼
Show Figures
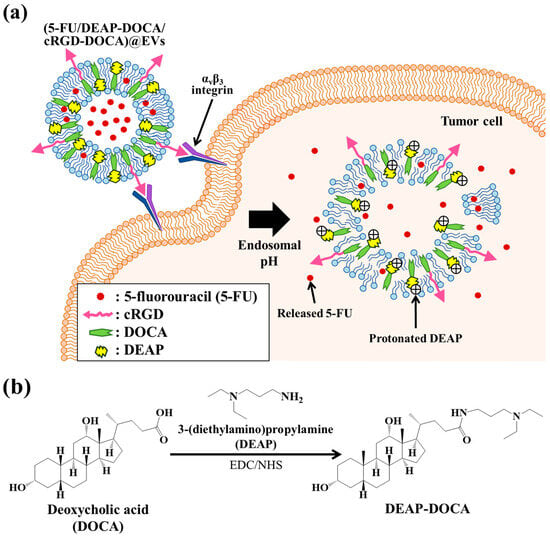
Figure 1
Open AccessArticle
A Two-Way Proposal for the Determination of Bioequivalence for Narrow Therapeutic Index Drugs in the European Union
by
Paulo Paixao, Alfredo Garcia Arieta, Nuno Silva, Zvonimir Petric, Milton Bonelli, José Augusto Guimarães Morais, Kevin Blake and Luís Filipe Gouveia
Pharmaceutics 2024, 16(5), 598; https://doi.org/10.3390/pharmaceutics16050598 - 28 Apr 2024
Abstract
In the European Union, bioequivalence (BE) for narrow therapeutic index (NTI) drugs is currently demonstrated when the 90% confidence interval for the ratio of the population geometric means of the test and reference products for AUC, and in some cases for Cmax, falls
[...] Read more.
In the European Union, bioequivalence (BE) for narrow therapeutic index (NTI) drugs is currently demonstrated when the 90% confidence interval for the ratio of the population geometric means of the test and reference products for AUC, and in some cases for Cmax, falls within the acceptance range of 90.00% to 111.11%. However, meeting this requirement results in an increased difficulty of demonstrating BE and a need for clinical trials with larger subject sample sizes, especially for medium-to-high variability drugs. To address this challenge, a scaled average BE based on the reference product within-subject variability for narrowing the acceptance range of NTI drugs was recently proposed. However, this approach showed increased type I error (T1E), especially close to the cut-off point between the unscaled and scaled portions of the method. Based on simulations, this limitation can be overcome by predefining the protocol the path to be followed: either the fixed 90.00–111.11% acceptance range approach or the previously proposed scaled average BE approach with a slight adjustment of the one-sided significance level α to 0.042 for a 2 × 3 × 3 partial replicate design and without a lower cut-off point. This results in a mixed approach allowing to reduce the sample size whilst not inflating the T1E.
Full article
(This article belongs to the Section Clinical Pharmaceutics)
►▼
Show Figures
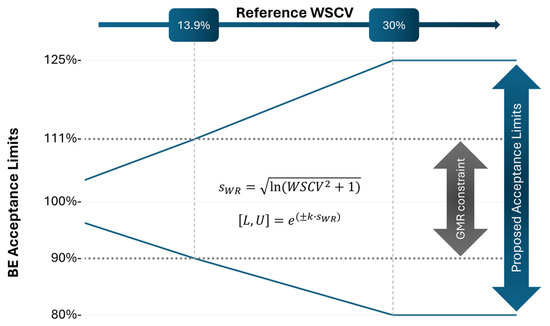
Figure 1
Open AccessArticle
An Effective Modification Strategy to Build Multifunctional Peptides Based on a Trypsin Inhibitory Peptide of the Kunitz Family
by
Ying Wang, Daning Shi, Wanchen Zou, Yangyang Jiang, Tao Wang, Xiaoling Chen, Chengbang Ma, Wei Li, Tianbao Chen, James F. Burrows, Lei Wang and Mei Zhou
Pharmaceutics 2024, 16(5), 597; https://doi.org/10.3390/pharmaceutics16050597 - 27 Apr 2024
Abstract
Peptides with antimicrobial activity or protease inhibitory activity are potential candidates to supplement traditional antibiotics or cancer chemotherapies. However, the potential of many peptides are limited by drawbacks such as cytotoxicity or susceptibility to hydrolysis. Therefore, strategies to modify the structure of promising
[...] Read more.
Peptides with antimicrobial activity or protease inhibitory activity are potential candidates to supplement traditional antibiotics or cancer chemotherapies. However, the potential of many peptides are limited by drawbacks such as cytotoxicity or susceptibility to hydrolysis. Therefore, strategies to modify the structure of promising peptides may represent an effective approach for developing more promising clinical candidates. In this study, the mature peptide OSTI−1949, a Kunitz-type inhibitor from Odorrana schmackeri, and four designed analogues were successfully synthesised. In contrast to the parent peptide, the analogues showed impressive multi-functionality including antimicrobial, anticancer, and trypsin inhibitory activities. In terms of safety, there were no obvious changes observed in the haemolytic activity at the highest tested concentration, and the analogue OSTI−2461 showed an increase in activity against cancer cell lines without cytotoxicity to normal cells (HaCaT). In summary, through structural modification of a natural Kunitz-type peptide, the biological activity of analogues was improved whilst retaining low cytotoxicity. The strategy of helicity enhancement by forming an artificial α-helix and ß-sheet structure provides a promising way to develop original bioactive peptides for clinical therapeutics.
Full article
(This article belongs to the Special Issue Advances in Bioactive Peptides from Natural Sources: Characterization, Biological Targets and Promising Applications)
Open AccessArticle
Targeted Thrombolysis with Magnetic Nanotherapeutics: A Translational Assessment
by
Ming-Lu Lin, Siao-Yun Wu, Jyh-Ping Chen, Yi-Ching Lu, Shih-Ming Jung, Shiaw-Pyng Wey, Tony Wu and Yunn-Hwa Ma
Pharmaceutics 2024, 16(5), 596; https://doi.org/10.3390/pharmaceutics16050596 - 27 Apr 2024
Abstract
Plasminogen activators, such as recombinant tissue-type plasminogen activators (rtPAs), while effective in treating thromboembolic diseases, often induce hemorrhagic complications due to non-specific enzyme activities in the systemic circulation. This study evaluated the targeting efficiency, efficacy, biodistribution, and potential toxicity of a rtPA covalently
[...] Read more.
Plasminogen activators, such as recombinant tissue-type plasminogen activators (rtPAs), while effective in treating thromboembolic diseases, often induce hemorrhagic complications due to non-specific enzyme activities in the systemic circulation. This study evaluated the targeting efficiency, efficacy, biodistribution, and potential toxicity of a rtPA covalently attached to chitosan-coated magnetic nanoparticles (chitosan-MNP-rtPA). The thrombolytic activity of a chitosan-MNP-rtPA was preserved by protection from an endogenous plasminogen activator inhibitor-1 (PAI-1) in whole blood and after circulation in vivo, as examined by thromboelastometry. Single-photon emission computed tomography (SPECT) demonstrated real-time retention of a 99mTc-MNP-rtPA induced by magnet application in a rat embolic model; an 80% reduction in rtPA dosage for a chitosan-MNP-rtPA with magnetic guidance was shown to restore blood flow. After treatment, iron deposition was observed in the reticuloendothelial systems, with portal edema and neutrophil infiltration in the liver at a ten-fold higher dose but not the regular dose. Nevertheless, no liver or renal toxicity was observed at this higher dose. In conclusion, the liver may still be the major deposit site of rtPA nanocomposites after targeted delivery; chitosan-coated MNPs are potentially amenable to target therapeutics with parenteral administration.
Full article
(This article belongs to the Special Issue Recent Advances in Biomedical Applications of Magnetic Nanomaterials)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Development and Efficacy Evaluation of Innovative Cosmetic Formulations with Caryocar brasiliense Fruit Pulp Oil Encapsulated in Freeze-Dried Liposomes
by
Letícia Kakuda, Patrícia M. B. G. Maia Campos and Wanderley P. Oliveira
Pharmaceutics 2024, 16(5), 595; https://doi.org/10.3390/pharmaceutics16050595 - 27 Apr 2024
Abstract
Encapsulation and drying technologies allow the engineering of innovative raw materials from plant biodiversity, with potential applications in pharmaceutical and cosmetic fields. Lipid-based nanoencapsulation stands out for its efficiency, ease of production, and versatility in encapsulating substances, whether hydrophilic or lipophilic. This work
[...] Read more.
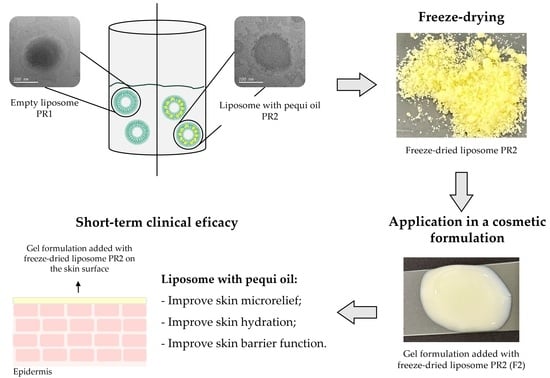
Encapsulation and drying technologies allow the engineering of innovative raw materials from plant biodiversity, with potential applications in pharmaceutical and cosmetic fields. Lipid-based nanoencapsulation stands out for its efficiency, ease of production, and versatility in encapsulating substances, whether hydrophilic or lipophilic. This work aimed at encapsulating pequi oil in liposomes and freeze-dried liposomes to enhance its stability and functional benefits, such as skin hydration and anti-aging effects, for use in innovative cosmetic formulations. Pequi oil—extracted from the Caryocar brasiliense fruit pulp, a plant species from Brazilian plant biodiversity—is rich in secondary metabolites and fatty acids. Liposomes and dried liposomes offer controlled production processes and seamless integration into cosmetic formulations. The physicochemical analysis of the developed liposomes confirmed that the formulations are homogeneous and electrokinetically stable, as evidenced by consistent particle size distribution and zeta potential values, respectively. The gel-type formulations loaded with the dried liposomes exhibit enhanced skin hydration, improved barrier function, and refined microrelief, indicating improvements in skin conditions. These results highlight the potential of dried liposomes containing pequi oil for the development of innovative cosmeceutical products. This research contributes to the valorization of Brazilian biodiversity by presenting an innovative approach to leveraging the dermatological benefits of pequi oil in cosmetic applications.
Full article
(This article belongs to the Special Issue Spray Drying and Encapsulation of Pharmaceuticals and Phytopharmaceuticals)
►▼
Show Figures
Graphical abstract

Journal Menu
► ▼ Journal Menu-
- Pharmaceutics Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Bioengineering, Biomolecules, Cancers, Diseases, Nanomaterials, Pharmaceutics
Dynamic Nano-Biomaterials in Tissue Regeneration and Cancer Therapies
Topic Editors: Ramar Thangam, Heemin Kang, Bibin G. Anand, Ramachandran Vijayan, Venugopal KrishnanDeadline: 15 May 2024
Topic in
Cancers, Cells, Diseases, Nanomaterials, Pharmaceutics
Findings, Insights and Perspectives on Central Nervous System Tumors
Topic Editors: João Basso, Carla Vitorino, Rui Vitorino, Ana FortunaDeadline: 31 May 2024
Topic in
Cancers, Cells, JCM, Radiation, Pharmaceutics, Applied Sciences, Nanomaterials, Current Oncology
Innovative Radiation Therapies
Topic Editors: Gérard Baldacchino, Eric Deutsch, Marie Dutreix, Sandrine Lacombe, Erika Porcel, Charlotte Robert, Emmanuelle Bourneuf, João Santos Sousa, Aurélien de la LandeDeadline: 30 June 2024
Topic in
Antibiotics, JPM, Pharmaceuticals, Pharmaceutics
Pharmacokinetic and Pharmacodynamic Modelling in Drug Discovery and Development
Topic Editors: Inaki F. Troconiz, Victor Mangas Sanjuán, Maria Garcia-Cremades MiraDeadline: 31 August 2024

Conferences
Special Issues
Special Issue in
Pharmaceutics
Exosomes as Drug Carriers for Cancer Therapy
Guest Editors: Sveva Bollini, Beáta SoltészDeadline: 10 May 2024
Special Issue in
Pharmaceutics
New Properties of Supramolecular Complexes and Drug Nanoparticles
Guest Editors: Elena Uspenskaya, Anton SyroeshkinDeadline: 20 May 2024
Special Issue in
Pharmaceutics
Novel Therapeutic Strategies for Glioblastoma
Guest Editors: Milica Pešić, Ana Podolski-Renić, Jelena DinićDeadline: 31 May 2024
Special Issue in
Pharmaceutics
Supramolecular Systems for Gene and Drug Delivery, 2nd Edition
Guest Editors: Francisco José Ostos, José Antonio Lebrón, Pilar López-CornejoDeadline: 10 June 2024
Topical Collections
Topical Collection in
Pharmaceutics
Feature Papers in Pharmaceutical Technology
Collection Editor: Thierry Vandamme
Topical Collection in
Pharmaceutics
Advanced Pharmaceutical Science and Technology in Korea
Collection Editors: Hyo-Kyung Han, Beom-Jin Lee
Topical Collection in
Pharmaceutics
Advanced Pharmaceutical Science and Technology in Estonia
Collection Editors: Karin Kogermann, Jana Lass
Topical Collection in
Pharmaceutics
Women in Pharmaceutics
Collection Editors: Donatella Paolino, Cinzia Anna Ventura